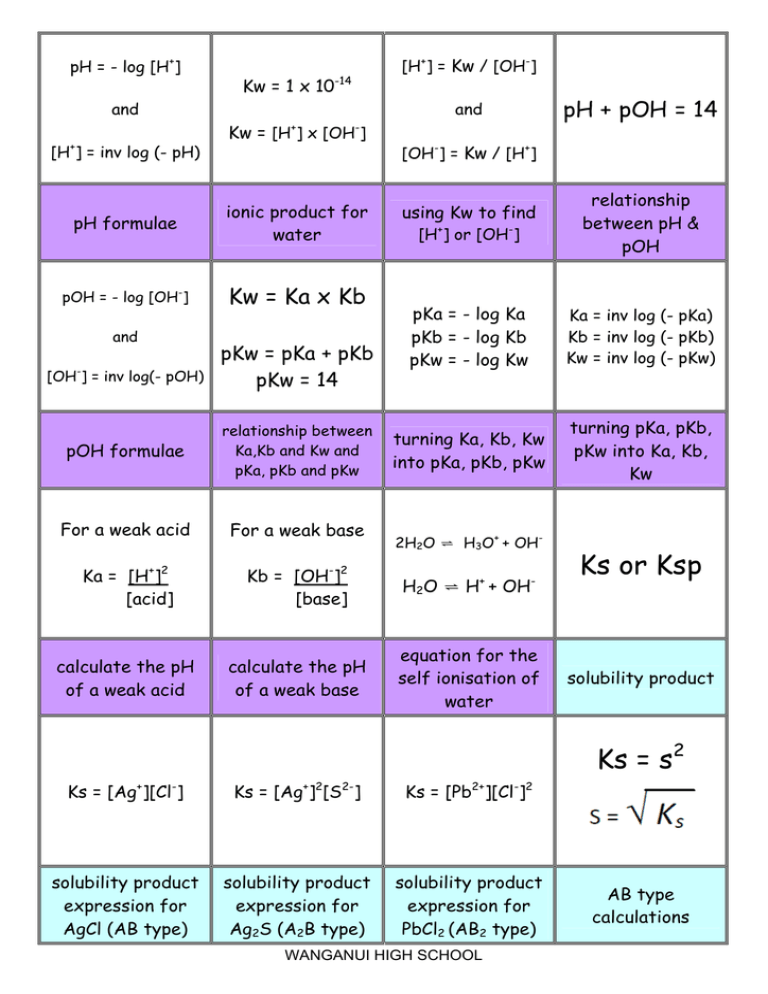
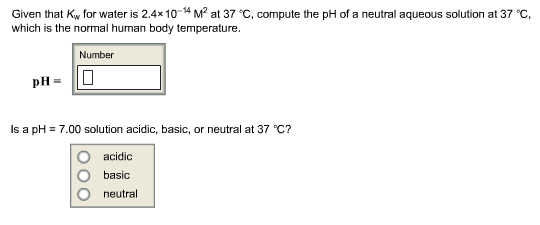
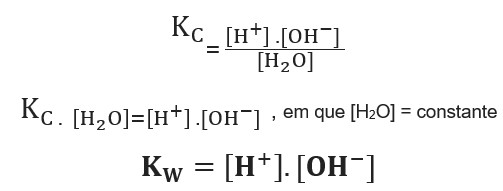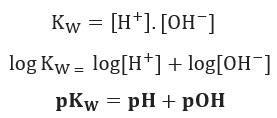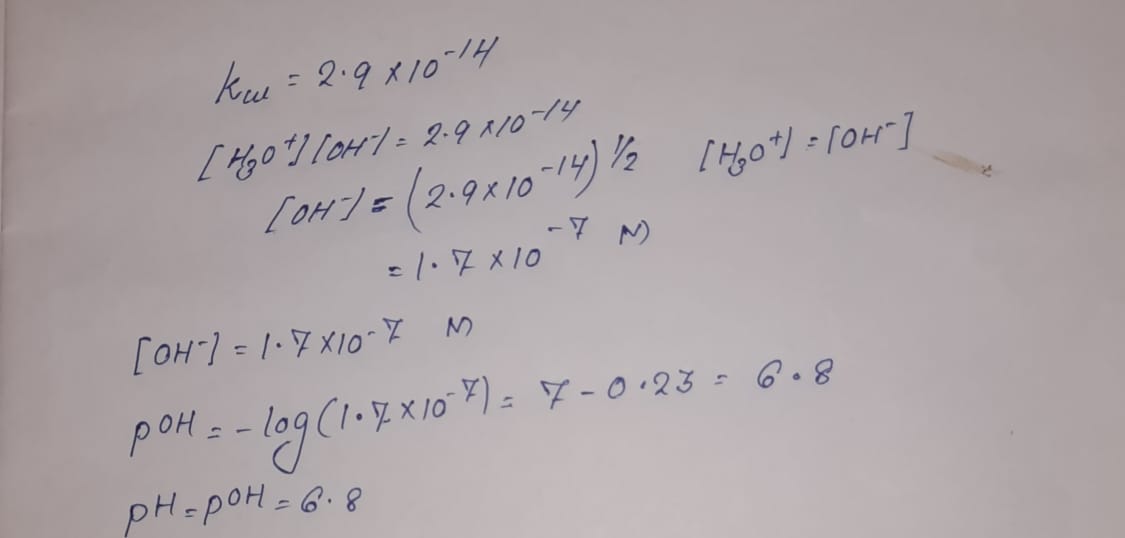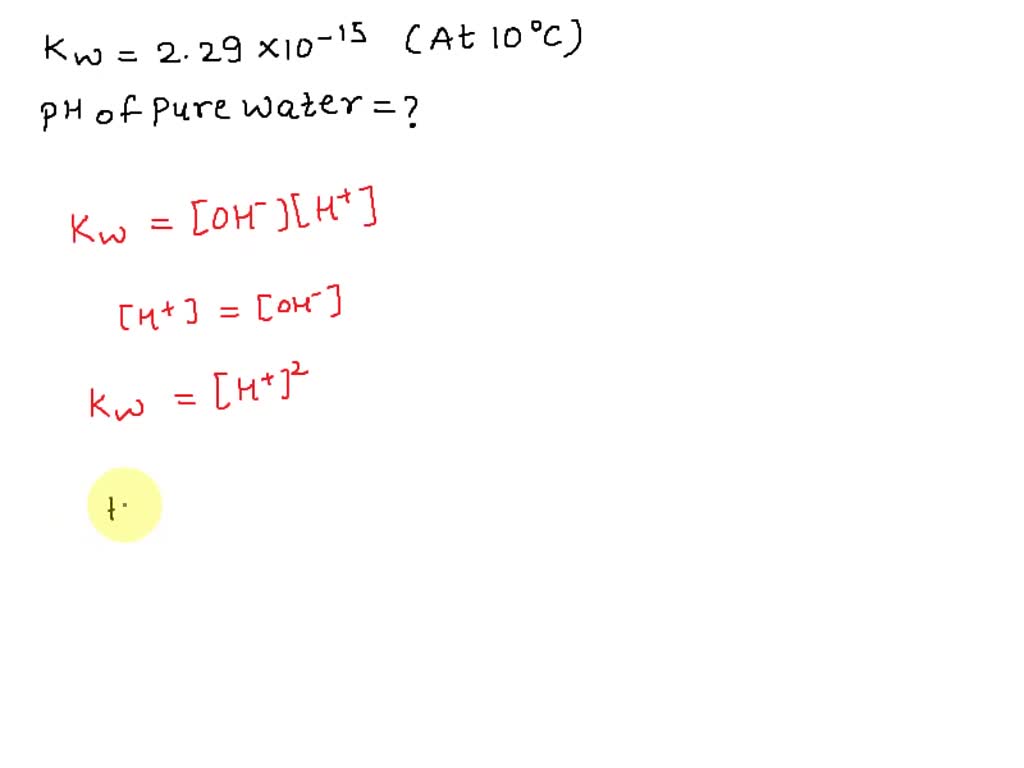pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems - YouTube
The ionization constant for water (Kw) is 9.311 × 10−14 at 60 °C. What is the [H3O+], [OH−], pH, and pOH for pure water at 60 °C? Thanks. - TopScience - Quora
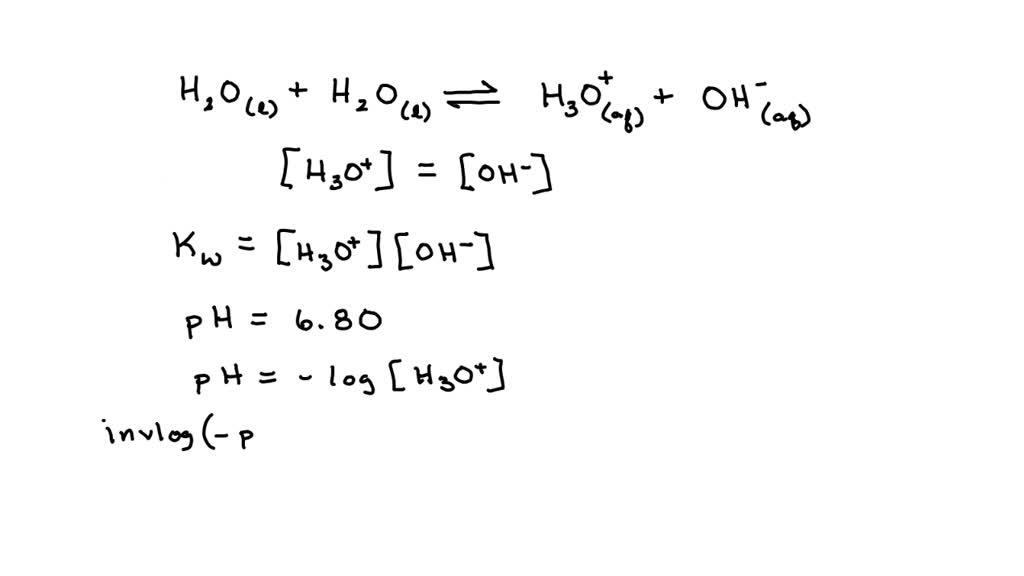
SOLVED: If the pH of pure water is 6.80 at 37 C, determine the value of KW at this temperature (it would be more relevant in medical applications that the value at 25 C).
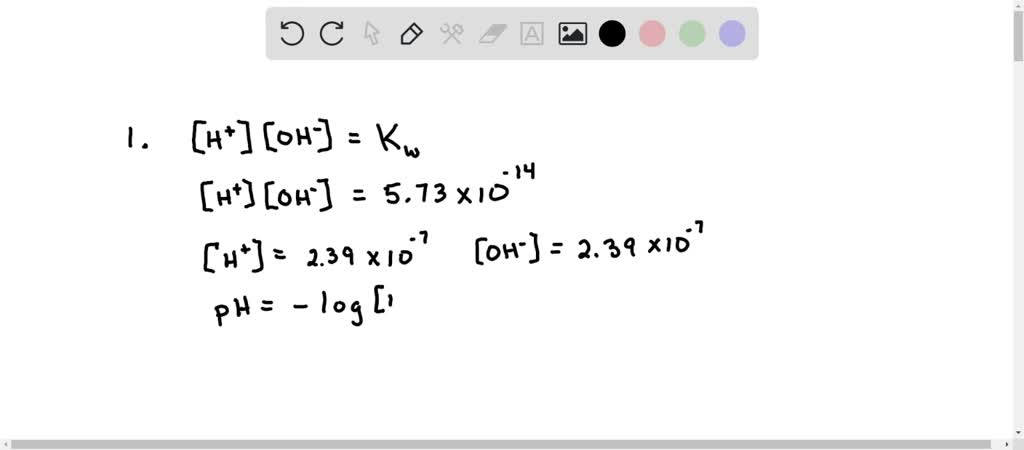
SOLVED: At 323 K, the value for Kw changes and is found to be 5.73 x 10 -14 . (i) Calculate the pH of water at this new, higher temperature. (1) (ii)

AutoIonization of Water, Ion Product Constant - Kw, Calculating H3O+, OH-, and pH Using Ice Tables - YouTube






![Acids and Bases Part 4: Kw and Calculation of [H+] and [OH-] - YouTube Acids and Bases Part 4: Kw and Calculation of [H+] and [OH-] - YouTube](https://i.ytimg.com/vi/IvP_PxetNUw/maxresdefault.jpg)