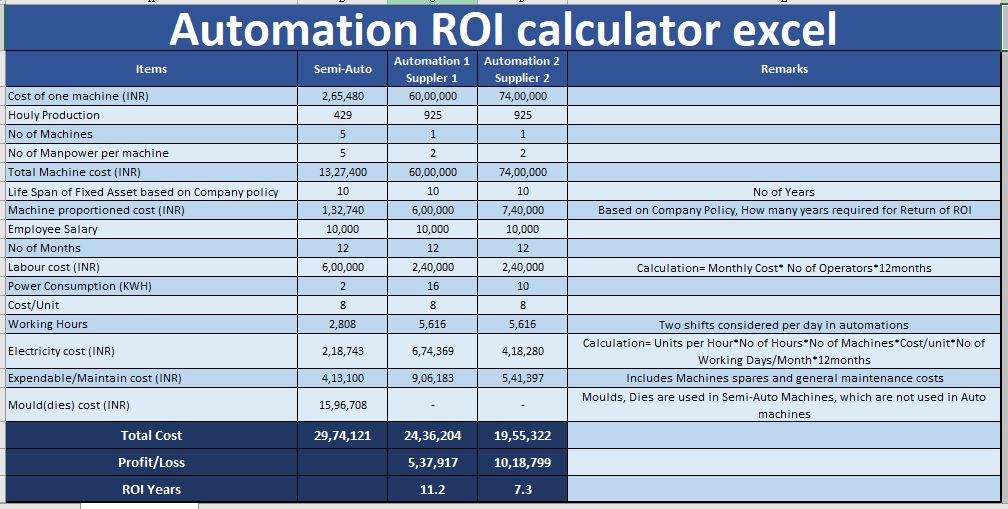
Calculate the percentage yield of the reaction 64g NaBH, with iodine produced 15.0 g of BIZ. NaBH, +412- B13 + Nal +4HI (At, mass, Na= 23, B = 10.8, I= 127) to

Texas Instruments TI-84 Plus CE Pink Graphing Calculator: Amazon.co.uk: Stationery & Office Supplies

Texas Instruments TI84PLUS - Graphing Calculator USB technology : Amazon.de: Stationery & Office Supplies

SOLVED: For the following reaction, 52.3 grams of iron(II) chloride are allowed to react with 127 grams of silver nitrate: iron(II) chloride(aq) + silver nitrate(aq) â†' iron(II) nitrate(aq) + silver chloride(s) What