Pilot single-arm study to investigate the efficacy and safety of endovenous Microwave ablations for treatment of varicose veins in Singapore – one year results of the MAESTRO registry - Tjun Yip Tang,

EU Regulatory Pathways for ATMPs: Standard, Accelerated and Adaptive Pathways to Marketing Authorisation: Molecular Therapy Methods & Clinical Development
CONSORT flow chart of the prospective cohort study. EMA ¼ endovenous... | Download Scientific Diagram

Effect of body size and posture on limb EMA. (A) Hindlimb EMA scaling... | Download Scientific Diagram

CONSORT flow chart of the prospective cohort study. EMA ¼ endovenous... | Download Scientific Diagram

Mutual recognition in the European system: A blueprint for increasing access to medicines? - ScienceDirect

EU Regulatory Pathways for ATMPs: Standard, Accelerated and Adaptive Pathways to Marketing Authorisation - ScienceDirect

Comparison of regulatory pathways for the approval of advanced therapies in the European Union and the United States - Cytotherapy
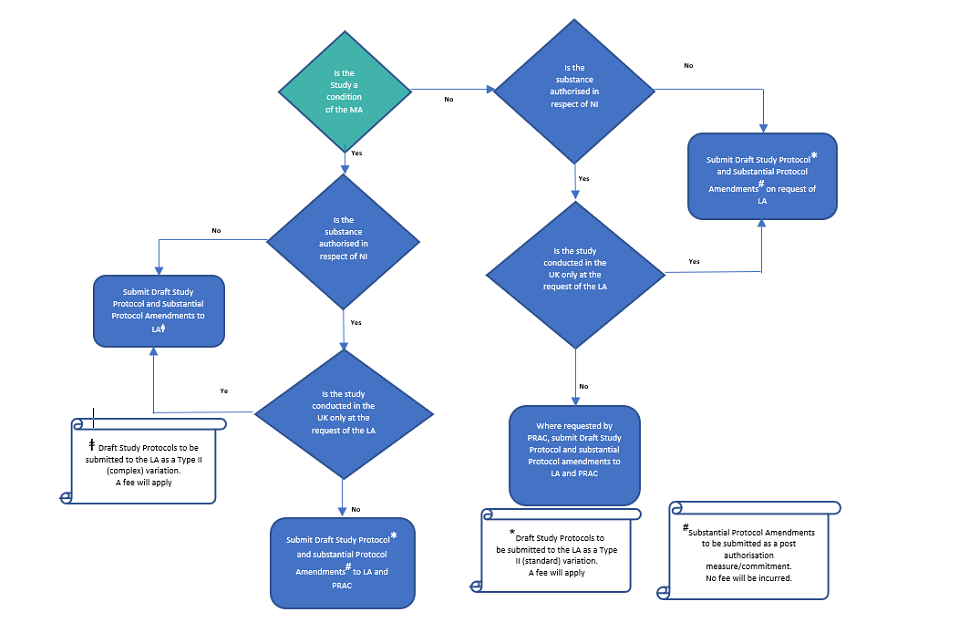
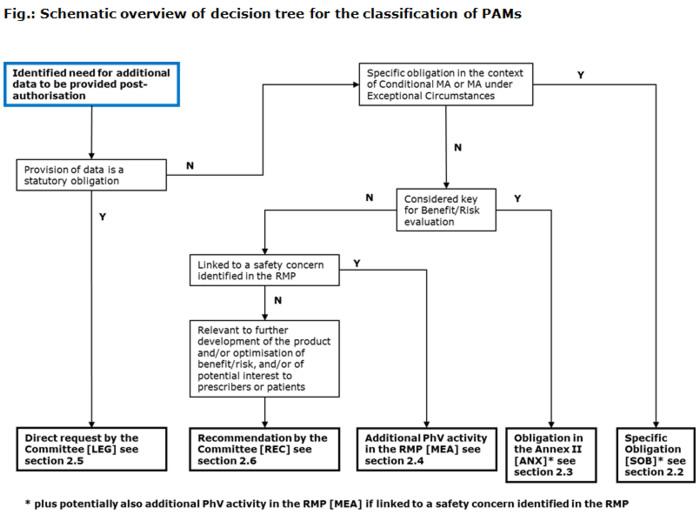
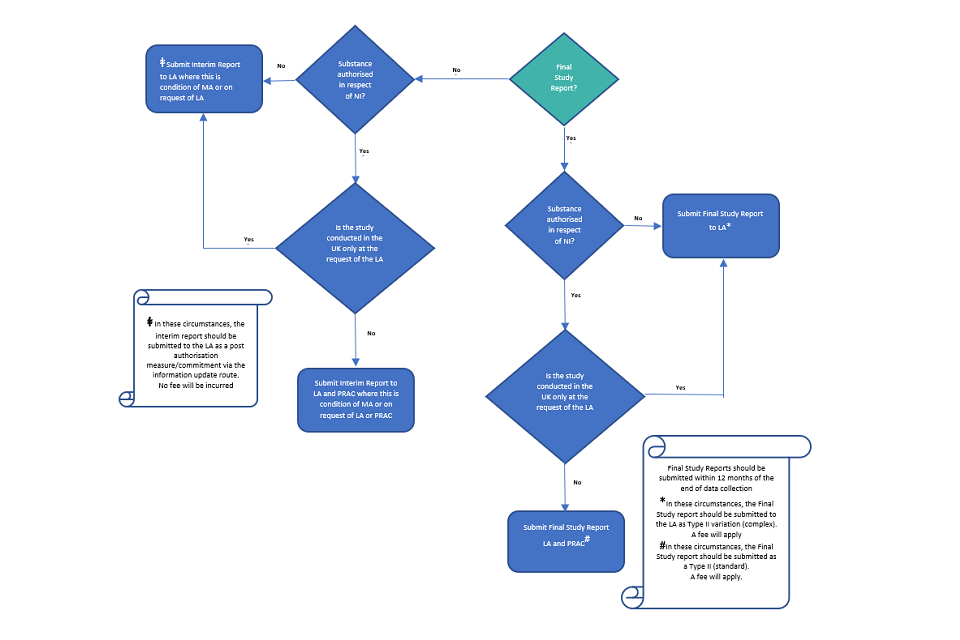
European Medicines Agency post-authorisation procedural advice for users of the centralised procedure

Frontiers | A Regulatory Risk-Based Approach to ATMP/CGT Development: Integrating Scientific Challenges With Current Regulatory Expectations